Acid Rain
Pure rainfall is slightly acidic, pH 5.6, because water reacts with atmospheric carbon dioxide to produce weak carbonic acid. When higher than normal amounts of nitric and sulfuric acid occur in the atmosphere, the result is precipitation with a pH below 5.6 which is referred to as acid rain. Acid rain includes both wet deposition (rainfall, snow, fog) and dry deposition (particulates). Acid rain formation result from both natural sources, such as volcanoes and decaying vegetation, and man-made sources, primarily emissions of sulfur dioxide (SO2) and nitrogen oxides (NOx) resulting from fossil fuel combustion. In the United States, roughly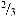 of all SO2 and
of all SO2 and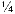 of all NOx come from electric power generation that relies on burning fossil fuels, like coal. Acid rain occurs when these gases react in the atmosphere with water, oxygen, and other chemicals to form various acidic compounds (Figure 6.12). The result is a mild solution of sulfuric acid and nitric acid. When sulfur dioxide and nitrogen oxides are released from power plants and other sources, prevailing winds blow these compounds across state and national borders, sometimes over hundreds of miles. Regions of greatest acidification tend to be downwind from heavily industrialized source areas of pollution.
of all NOx come from electric power generation that relies on burning fossil fuels, like coal. Acid rain occurs when these gases react in the atmosphere with water, oxygen, and other chemicals to form various acidic compounds (Figure 6.12). The result is a mild solution of sulfuric acid and nitric acid. When sulfur dioxide and nitrogen oxides are released from power plants and other sources, prevailing winds blow these compounds across state and national borders, sometimes over hundreds of miles. Regions of greatest acidification tend to be downwind from heavily industrialized source areas of pollution.
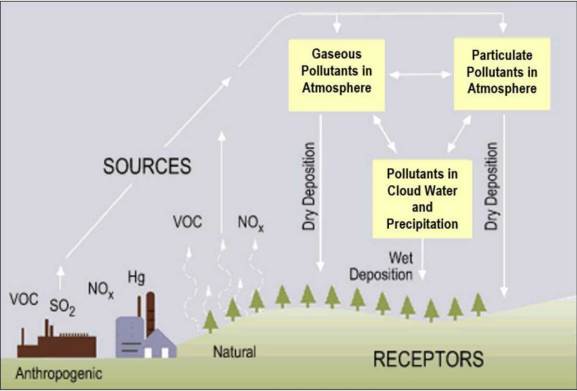
Acid rain is a serious environmental problem that is particularly damaging to lakes, streams, and forests and the plants and animals that live in these ecosystems, as well as to infrastructure. Some of the impacts include:
• Leaching of soil nutrients such as calcium, magnesium, and potassium out of the topsoil, altering soil chemistry and harming plants and soil organisms.
• Acid rain causes the release of substances that are toxic to trees and other plants, such as aluminum, into the soil. Scientists hypothesize that this combination of loss of soil nutrients and increase of toxic aluminum may be one way that acid rain harms trees (Figure 6.13). Such substances also wash away in the runoff and are carried into streams, rivers, and lakes.
• Damage to automotive paints and other coatings. The reported damage typically occurs on horizontal surfaces and appears as irregularly shaped, permanently etched areas.
• Acidic particles contribute to the corrosion of metals (such as bronze) and the deterioration of paint and stone (such as marble and limestone). These effects significantly reduce the societal value of buildings, bridges, cultural objects (such as statues, monuments, and tombstones) (Figure 6.14).
• Sulfates and nitrates that form in the atmosphere contribute to visibility impairment, meaning we cannot see as far or as clearly through the air. Sulfate particles account for 50 to 70 percent of the visibility reduction in the eastern part of the U.S., affecting our enjoyment of national parks, such as the Shenandoah and the Great Smoky Mountains.


Attribution
Zehnder, Caralyn; Manoylov, Kalina; Mutiti, Samuel; Mutiti, Christine; VandeVoort, Allison; and Bennett, Donna, “Introduction to Environmental Science: 2nd Edition” (2018). Biological Sciences Open Textbooks. 4.
https://oer.galileo.usg.edu/biology-textbooks/4
This work is licensed under a Creative Commons Attribution-Noncommercial-Share Alike 4.0 License.

