4 Vaccines and Vaccinations: Production Animals
Learning Objectives
- Describe risks and management factors to consider when vaccinating cattle, small ruminants, and swine
- Describe cause and clinical manifestations of conditions for which we vaccinate cattle, small ruminants, and swine
- Explain where and how to vaccinate production animals
- Define what is meant by commercial, autogenous, and on-farm vaccine production
- Create appropriate vaccine protocols for cattle, small ruminants, and swine
*
 Dairy Cattle
Dairy Cattle
Dairy and beef cattle generally always live in a herd. Management practices will therefore affect multiple animals. Likewise, from an infectious disease standpoint, it has to be kept in mind that not one, but multiple animals will be exposed to a particular pathogen. As a result, the prevention of disease on dairy farms needs to be targeted towards groups of animals and the entire herd rather than individual animals. Disease prevention is crucial to allow the economic survival of the farm and to ensure animal welfare. Specific brands of vaccines will not be discussed for beef or dairy cattle because there is tremendous variability in vaccines used. Examples of diseases for which cattle may be vaccinated include bovine viral diarrhea (BVD), infectious bovine rhinotracheitis (IBR), parainfluenza (PI3), bovine respiratory syncytial virus (BRSV), leptospirosis, Pasteurella, Histophilus (Hemophilus), Campylobacter (Vibrio), and clostridial diseases. Specifics of vaccines and their use will be covered in species-specific coursework later in the curriculum. An example of which vaccines are used at which life stage is included in the section on beef cattle as are descriptions of the diseases we protect against with vaccination in cattle.
Risks and Management Factors
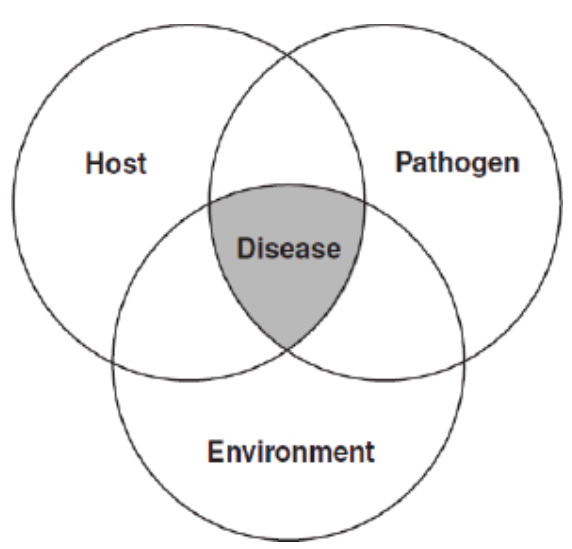
Factors to Consider in Disease Prevention
Diseases occur due to multiple factors. Their relationship is sometimes called the infection triangle.All three factors (environment (e.g. management practices), host, and pathogen) are important for the development or prevention of disease and all three need to be considered during disease prevention.
The host has various defense mechanisms to prevent or to deal with infections. A given species or animal might not be susceptible to a certain disease. For example, E. coli O157:H7, a potentially fatal bacterium for humans, is a normal gut commensal for cattle. Cattle do not have the receptors for the toxins of this bacterium in their kidneys and so they cannot develop the hemolytic uremia syndrome that affects humans. Other defense mechanisms, specific and non-specific, are part of the immune system. These defense mechanisms may be impaired due to the animal’s age, inadequate nutrition, and other stressors from within the animal, or may be impaired due to management practices. Stress due to heat, weaning, malnutrition, infection pressure (amount of exposure to a given pathogen), transport, and other factors will negatively impact how the immune system can react to a “pathogen attack”.
Furthermore, an infectious disease can only spread through a population if the pathogen ‘finds’ susceptible animals to infect. Some animals might get sick, but if a population (herd) has many resistant animals, the probability of new infections can be so low that the disease ‘dies out’ even if not all animals are resistant to the disease. This is called “herd immunity”. In human medicine this principle is used every year as we receive flu shots. The aim is to reduce the number of susceptible humans through flu vaccinations and so reduce the likelihood of new infections, particularly in children and immunocompromised people.
So for obvious reasons, the environment and management practices play a crucial role in disease prevention on farm by influencing the animal and the pathogen. Any management practices that decrease the infection pressure on animals (for example, hygiene or biosecurity) will support animals in coping with infection pressure and will reduce the likelihood that animals will even get exposed to the pathogens.
Vaccination is only part of this puzzle and cannot be used as a standalone practice to prevent disease or infection on farm. Vaccination is not a miracle intervention. It does not result in immediate immunity or resistance against diseases in all vaccinated animals. First, it takes time for the immune system of the animal to react to the vaccine and second, just because an animal is vaccinated, it does not automatically mean that the animal cannot be infected or develop the disease. However, vaccination can be used as one of many management practices to help animals cope with potential infection threats.
Vaccinating Production Animals
When thinking about vaccinating a dairy herd, you have to understand the production system and the diseases that are of importance for that particular dairy farm. You need to ask several questions:
- What management practices are already in use on this dairy farm and where could improvements possibly be made to decrease infection pressure? Examples include improving hygiene, not bringing in animals from unknown sources, and limiting foot traffic between age groups.
- Which pathogens are of concern for their impact on the health and production of animals? If an animal is unlikely to ever encounter a pathogen, it is probably not necessary (and is possibly too costly) to vaccinate against it. Do not over-vaccinate animals, and tailor the vaccine program toward each herd individually.
- Do benefits of a possible vaccination outweigh the costs of the disease or could vaccination be economically detrimental? For example, Foot-and-Mouth-Disease (FMD) has not been diagnosed in the United States since the 1920s and the detection of positive antibody titers in animals would result in serious trade restrictions. Therefore, routine vaccination against FMD is probably not economically sound.
- When is the best time to vaccinate the animals? On farms, it is always groups of animals, not individuals, that are vaccinated with a given vaccine. The timing of the vaccination has to take into account which age groups are at the highest risk for diseases, how long it takes for an animal to build up immunity, when the animals are going to be handled for any reason, and when the potential stressors occur in an animal’s life. For example, when is the best time to vaccinate a dairy cow against pathogens associated with calfhood diarrhea? Calves desperately need to ingest the antibodies in the colostrum of its dam (passive immunization) to allow them to handle infections until they were able to build their own immune response. The cow needs several weeks after vaccination to develop significant antibody concentrations that can be deposited into colostrum. So, keeping biology and management practices in mind, in this example, it is best to vaccinate the dam in the weeks before calving so the dam can deliver these antibodies through the colostrum to the calf to prepare the calf’s immune system for a potential exposure in the first week of life.
- Read the label of the vaccines! What is the dosage and route for vaccine administration? How long will the immunity last? Some vaccines are for heifers only and some must not be given during pregnancy. Other vaccines (in particular those against zoonotic pathogens) are to be administered by the veterinarian only. Additionally, you have to remember that you are administering a biological drug to a food production animal. Therefore, you need to consider withdrawal times to ensure safety of meat and milk from that animal as products from that animal may enter the human food chain. Withdrawal times may be based on the organism itself or on other components of the vaccine (for example, preservatives or adjuvants).

*
 Beef Cattle
Beef Cattle
The role of vaccines in a preventative health program for beef cattle is to prevent or eliminate clinical disease in an individual or a population of cattle. This is accomplished by decreasing the incidence and severity of disease through increased level of immunity as part of a preventive health program. No vaccine is 100% effective. Vaccines are part of a preventive health program that should include management procedures and handling, parasite control, and nutritional considerations that reflect an in-depth understanding of not only the beef production system but also farm-specific issues and goals.
Factors to Consider in Developing a Vaccination Strategy
When developing a vaccination strategy, several factors must be considered. Some of these factors are:
- Class of cattle (age)
- Nursing calves, weaned calves, yearling cattle, replacement heifers, pregnant cows, open cows, breeding bulls
- Production system the cattle are in
- Cow-Calf, Stocker/Backgrounder, Feedlot
- Open system (purchasing and bringing in cattle) vs. Closed system
- What to vaccinate for
- Reproductive Diseases
- Respiratory Diseases
- Systemic Diseases
- Goals of the producer / history
- Future plans, direction, etc.
- Previous challenges/approaches
- Risk tolerance of the producer
- High vs. Low
- Expectations
- Facility availability / husbandry skills
- Ease of handling, stress of handling
- Labor needed/available
- Compliance ability
Vaccination does not equal immunization, so designing the vaccine program also requires consideration of factors affecting immune response. These factors can include:
- Age of calf
- The bovine’s immune system begins to develop prior to birth but does not reach maximum responsiveness until later in life.
- In addition there is thought to be “maternal interference” where passive immunity obtained through colostrum consumption may interfere with ability for young calves to respond to some vaccines.
- Nutritional Status
- Current and previous nutritional status should be considered. Cattle that have been deficient in key components (protein, energy, vitamins, and minerals) may have a decreased ability to mount an immune response.
- Stressors
- Vaccines should be administered at times of low or minimized stress and, if possible, prior to expected stressors. Common stressors include environmental temperature, disease status, parasite load, transport, weaning, nutritional changes, handling, management practices (castration, dehorning, etc), and comingling.
- Timing of vaccination
- The time between vaccination and challenge by infectious agents is important. If using a killed vaccine, you need adequate time for a booster dose when first administering.
- Additional considerations such as time following parturition or prior to breeding, colostrum production, pregnancy status, and avoiding stressors are important as well.
- Try to incorporate vaccination with other herd management procedures when possible to take full advantage of handling efficiencies; your goal is to minimize frequency of handling, which is a stressor.

Recommended Vaccination Schedules
Preventive health programs for beef cattle in the upper Midwest provide recommendations for vaccines at specific lifestages.
Calves
| LIFESTAGE | RECOMMENDED VACCINE |  |
| Birth |
|
|
| Branding (mid summer) or Prebreeding |
|
|
| Preweaning |
|
|
| Weaning |
|
|
| At entry to feedlot (Variable; based on vaccine history and risk class) |
|
|
| Revaccination (Variable; based on vaccine history and risk class) |
|
Beef Replacement Heifers
| LIFESTAGE | RECOMMENDED VACCINES |  |
|---|---|---|
| Prebreeding 1 (8-10 months) |
|
|
| Prebreeding 2 (30 days before breeding) |
|
|
| Precalving 1 if on a fall (pregnant cow) vaccine schedule |
|
|
| Precalving 2 (5-7 weeks precalving) |
|
|
| Move to cow schedule |
Adult Cows and Bulls
| LIFESTAGE | RECOMMENDED VACCINES |  |
| Fall Pregnancy Check or Prebreeding |
|
|
| Precalving (3-7 weeks precalving) |
|
Vaccination Risks and Methods
As in small animals, side-effects can occur with vaccination in cattle. These include soreness/swelling/knots at the injection site, fever, off-feed, and potentially anaphylaxis (allergic reactions). “Endotoxin stacking,” the act of giving multiple killed bacterin vaccines (often Gram negative or Gram negative-like) vaccines together, has been associated with higher incidence of adverse reactions, failed immune response, and death.To minimize vaccine risks:
- Avoid the administration of more than two killed bacterin vaccines at the same time
- Do not shake killed bacterial vaccine such as clostridial (black-leg) products
- Never vaccinate on days where cattle are heat stressed
- Store vaccines properly
- 35-45oF, DO NOT FREEZE, and avoid sunlight
- Administer with proper care
- Only use licensed, quality vaccines
Beef Quality Assurance Vaccination Administration Guidelines
Any injection has the ability to cause a detectable lesion at slaughter. Beef Quality Assurance programs offer guidelines in proper administration of injectable products with the goal of maintaining and increasing the quality of the end product.Some general recommendations include:
- Use vaccines labeled for subcutaneous route of administration whenever possible.
- Administer all vaccines in the neck region.
- Inject in a clean site.
- Replace needles frequently or use single use needles.
- Use an appropriately sized needle for the intended injection route.
- Use good sanitation when withdrawing vaccines.
- Use properly cleaned and maintained syringes.
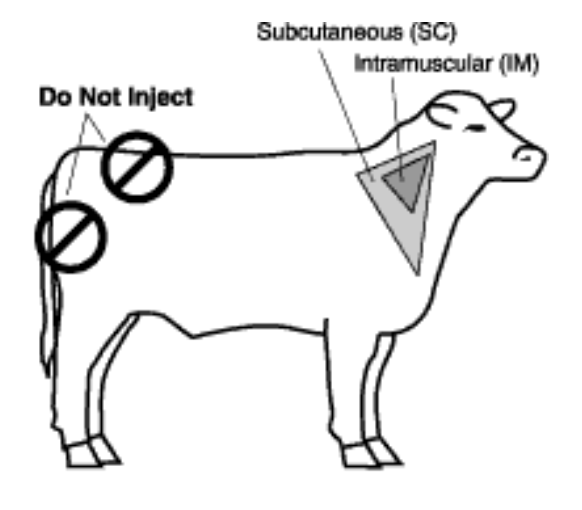
Route of Vaccine Administration in Cattle
| Subcutaneous (SC) (1/2 to 3/4 inch needle*) |
Intravenous (IV) (1 1/2 inch needle*) |
Intramuscular (IM) (1 to 1 1/2 inch needle*) |
|||||||
| Cattle Weight (lbs) | <300 | 300-700 | >700 | <300 | 300-700 | >700 | <300 | 300-700 | >700 |
| Gauge needle required for thin injectable viscosity (ex: saline) |
18 | 18-16 | 16 | 18-16 | 16 | 16-14 | 20-18 | 18-16 | 18-16 |
| Gauge needle required for thick injectable viscosity (ex: oxytetracycline) |
18-16 | 18-16 | 16 | 16 | 16-14 | 16-14 | 18 | 16 | 16 |
*Select the needle to fit the cattle size (the smallest practical size without bending)
—“Recommended needle size”, https://bqa.unl.edu/bqanebr-article-3
Diseases to Vaccinate Against in Beef and Dairy Cattle
Diseases in beef and dairy cattle to vaccinate against, include:
- Bovine viral diarrhea (BVD)
- Infectious bovine rhinotracheitis (IBR)
- Parainfluenza (PI3)
- Bovine respiratory disease complex
- Leptospirosis
- Campylobacter
- Clostridial diseases
- Brucellosis
Bovine Viral Diarrhea
BVD is a common cause of respiratory and reproductive issues in the herd. It is an economically important disease in many countries. The disease is caused by the bovine viral diarrhea virus, which may cause primary disease or be part of a large complex of infections. Cows infected during pregnancy may abort, give birth to a stillborn calf, or give birth to a live calf. Calves can be infected in utero or after birth. Calves born carrying BVD virus will never clear the infection and will shed the virus continuously into the farm environment. Signs of BVD in adults are highly variable and include fever, lethargy, loss of appetite, ocular discharge, nasal discharge, diarrhea, and decreased milk production. Infected calves may have cerebellar hypoplasia; signs of this are ataxia, tremors, a wide stance, and failure to nurse.
Infectious Bovine Rhinotracheitis
IBR is a highly contagious, infectious respiratory disease that is caused by bovine herpesvirus I. It can affect young and older cattle. IBR causes acute inflammation of the respiratory tract; clinical signs include those of respiratory disease (mucosal hyperemia, erosive mucosal lesions, nasal discharge, coughing), and also conjunctivitis, abortion, and encephalitis. After the first infection, the virus is sequestered in neurons in the brain as a latent infection. At times of stress, the virus undergoes recrudescence and can be shed from the eyes and nose. Because an infected animal never clears the virus, animals that are seropositive for bovine herpesvirus 1 cannot be shipped into countries that are free of the disease and cannot be housed in artificial insemination (AI) centers. Carrier cattle should be identified and removed from the herd.
Parainfluenza
The PI3 virus infects the upper respiratory mucosa, where it is shed in aerosols (sneezing and coughing) and by direct contact. PI3 causes only respiratory disease. In mature cattle, infection is mild. The most important consequence of PI3 infection in bovine respiratory disease is that it predisposes animals to concurrent infection with IBR or bacterial respiratory pathogens.
Bovine Respiratory Disease Complex
Bovine respiratory disease complex is caused by a variety of pathogens, including the three above. Other organisms against which cattle may be vaccinated to minimize this disease complex are bovine respiratory syncytial virus (BRSV) and bacteria including Pasteurella multocida, Mannheimia haemolytica, and Histophilus (formerly Hemophilus) somnus. These pathogens interact with each other to cause disease. Disease is worsened in the presence of stressors including parasites, weaning, change of feed, variation in ambient temperature and humidity, and weather. Clinical signs depend on age of the animal, organism(s) involved, and stage of the disease. Bovine respiratory disease is closely linked with fever; it is one of the most common causes of fever in cattle and fever may be the first sign of disease in affected cattle. Other signs include mental dullness, lack of appetite, rapid shallow breathing, and discharge from the nose and eyes (watery to purulent to bloody). Coughing will be mild and tentative early in the disease course and prominent (“honking”) later in the disease course.
Leptospirosis
Leptospirosis is a zoonotic disease, caused by bacteria of the genus Leptospira. There are many different serotypes of disease and prevalence varies with geographic location. Serovars include hardjo-bovis, pomona, canicola, icterohaemmorhagiae, and grippotyphosa. Maintenance hosts, also called reservoir hosts, carry the bacteria and are a source of exposure to other susceptible animals. Maintenance hosts for leptospirosis include cattle, pigs, dogs, raccoons, skunks, and rodents. Animals can be infected by serovars maintained by their own species (host-adapted infection) or by serovars maintained by other species (non-host-adapted infection). Leptospirosis can be transmitted either directly between animals or indirectly, through the environment. Cattle are the maintenance hosts for L. hardo-bovis; disease in cattle with this serovar is less severe but can have a significant economic impact. Cattle infected with other seorvars, especially L. pomona, suffer more severe illness. Clinical signs vary with the herd’s degree of resistance or immunity, the infecting serovar, and the age of the animal infected. Host-adapted infections in cattle can occur in animals of any age and mainly affect fertility and the renal system. Infected cattle may shed the organism in their urine for weeks to months. Non-host-adapted infection in cattle is typically due to L. pomona and mainly affects the hemolymphatic, urinary, and reproductive systems. Acute disease is characterized by hemolytic anemia, which causes red urine and jaundice. Reproductive effects include infertility, stillbirths, and abortion 1-3 months after infection. Lactating cows may suffer from mastitis, with decreased production and milk that is thick and yellow.
Campylobacter
Campylobacteriosis, formerly called Vibriosis, is caused by the bacterium Campylobacter fetus and is spread by infected bulls when they mate with susceptible cows and heifers. Once infected, a bull remains an asymptomatic carrier of the condition. Transmission is venereal; non-venereal transmission is unlikely to occur. When introduced to a herd the disease spreads rapidly because the cows and heifers have no immunity. Conception rates drop to around 40%. As immunity develops, the disease rate drops but reinfection often occurs as immunity wanes about a year after the initial infection. Conception rates in chronically infected herds are usually between 65 and 75%, with replacement heifers (newly introduced animals) being most severely affected. The infection can prevent implantation of a fertilized egg in the uterine lining, or more commonly, causes death of the developing embryo. When the embryo is lost, the cow goes back into heat and usually can be rebred successfully, since she has now developed immunity. Occasionally the disease results in permanent infertility.
Clostridial Diseases
Clostridial diseases strike cattle suddenly, often causing death before any clinical signs are seen. The bacteria that cause these diseases create very long-lived spores that are found everywhere in the environment and can easily be picked up by grazing cattle or enter the body through a wound. Bacteria may live in the gastrointestinal tract and spores also may be present in the tissues of healthy animals. Not all species of Clostridium cause disease but those that do usually are fatal. Examples include:
- C. septicum – malignant edema
- C. chauvoei – blackleg
- C. perfringens types A, B, C, and D – enterotoxemia
- C. tetani – tetanus
- C. botulinum – botulism
Contributing factors are necessary to allow the bacteria to multiply and cause disease. This may include injury or invasive procedures such as surgery, giving birth (parturition), or puncture wounds. Diet changes, overeating, and acidosis may permit clostridial organisms in the gut to multiply and cause disease. Clinical signs differ depending on the specific organism and may include sudden death in apparently healthy animals, lethargy or depression, high fever, anorexia, localized stiffness or muscle spasms, port wine colored urine, acute lameness and swelling in the hips and shoulders with a crackling sound when the skin is pressed (blackleg), or flaccid paralysis (botulism).
Brucellosis
Brucellosis is an infectious disease that spreads between animal species and between animals and humans. In cattle, the bacterium involved usually is Brucella abortus. Brucellosis is highly contagious, spreading very easily between cattle. The primary clinical sign is late-term abortion and the aborted calf, membranes, and fluids all contain large numbers of bacteria.

*
 Small Ruminants
Small Ruminants
Vaccination Methods
Vaccines cannot replace good management practices. When vaccinating sheep and goats, make sure they are healthy animals, and that the injection site is clean and dry to prevent introducing infection with your injection. Make sure you have adequate handling facilities such as a properly constructed chute or pen. When vaccinating multiple animals using an automatic syringe, verify that the correct dose is being administered. Needles should be changed every 12-20 animals (generally every pen or chute-full) and anytime the needle is dull, burred, or bent. Some facilities will be part of specific disease-control programs (caprine arthritis encephalitis virus (CAE) or ovine progressive pneumonia (OPP)); in that instance, multi-dose syringes may be used but a new needle must be used for each animal. In general, 18 gauge 5/8” needles are used for adult animals and 20 gauge 1/2 to 5/8” needles are used for young animals. Make sure all animals are individually identified (tags, tattoos) and that there is a system for record keeping so you know which animals received which vaccines.
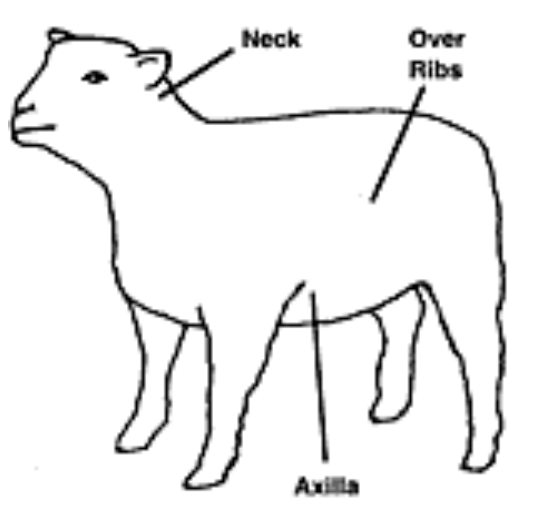
To vaccinate, part the wool if necessary, raise the skin to form a tent and insert the needle into the tent opening so that the needle is almost parallel with the neck. The site of injection is important if the animal you are vaccinating is to be used for food. Vaccines are given so as to minimize carcass and hide degradation due to abscess or scar formation. Vaccines should be given subcutaneously in the neck region or in the ventral aspect of the axillary space. The meat from these regions is of low value and pelt damage in these areas easily can be trimmed with minimal effect on value. Injection over the ribs is not optimal but it can be used if the animal cannot be restrained to allow you to use another location.
How to Vaccinate Sheep and Goats
 |
 |
*
Factors to Consider in Developing a Vaccination Strategy
Which vaccines are used depends on:
- History at the individual farm
- Age of the sheep or goats
- Previous disease problems
- Open or closed flock/herd status – are animals coming and going for shows, purchases, breeding?
- Geographic region
- Soil type
- Diet and flock/herd economics
- Withdrawal time (refer to the label!)
To choose vaccines, consider:
- Real and high-likelihood risks
- Since all ruminants are at risk for Clostridium perfringens type D, toxoid against this agent ideally should be used in all sheep flocks and goat herds
- Show animals have considerable exposure to contagious ecthyma virus (soremouth) and caseous lymphadenitis
- Open flocks have exposure to foot rot and abortion diseases
- All animals should be vaccinated with:
- CD-T vaccine – Clostridium perfringens is more commonly called enterotoxemia or overeating disease. Type C primarily affects lambs and kids during their first few weeks of life. Type D (pulpy kidney disease) affects lambs and kids that are usually over a month of age, particularly those that are creep-fed or finished on concentrate diets. Clostridium tetani is the causative agent of tetanus. CD-T toxoid is the vaccine usually used to protect healthy sheep and goats against these clostridial diseases.
- Most animals should be vaccinated against Campylobacter and Chlamydophila (abortion diseases) and contagious ecthyma (soremouth).
- Some animals should be vaccinated against contagious lymphadenitis and rabies and even more rarely, against E. coli, bluetongue virus, and Brucella ovis.
- Some people recommend use of cattle or equine vaccines in small ruminants but they are not labeled for this use and efficacy has not been well demonstrated.
Diseases in Sheep and Goats
Diseases to be vaccinated against in sheep and goats include:
- Clostridial diseases
- Abortion diseases in sheep
- Contagious ecthyma (soremouth, orf, scabby mouth or pustular dermatitis
- Caseous lymphadenitis (CLA, boils, abscesses or cheesy gland)

Clostridial Diseases
Clostridial organisms of various types are found in the soil, where they can survive for a very long time. Most clostridial organisms also survive naturally in the gastrointestinal tract of healthy animals. Sheep can be infected with various clostridial diseases (see diseases in cattle) but the most common are enterotoxemia types C and D and tetanus. Enterotoxemia type C (hemorrhagic enteritis or bloody scours) is caused by Clostridium perfringens type C and affects lambs during their first few weeks of life, causing a bloody infection of the small intestine. It is often related to indigestion and is predisposed by a sudden change in feed, for example an increase in the dam’s milk supply or beginning creep feeding as lambs are being weaned. Enterotoxemia type D (overeating disease or pulpy kidney disease) is caused by Clostridium perfringens type D and commonly strikes the largest, fastest growing lambs in the flock. It is caused by a sudden change in the feed that causes the organism, which is already present in the lamb’s gastrointestinal tract, to proliferate, causing a toxic reaction. It is most common in lambs that are on high concentrate rations but can also occur when lambs are nursing from dams that are heavy milkers. It usually affects lambs over one month of age. Sudden death may be the first sign. Other clinical manifestations include neurologic signs, seizures, and diarrhea. Tetanus is caused by Clostridium tetani, a soil inhabitant that is a prolific spore producer. This disease is usually related to tail docking and castration, although any wound can harbor the tetanus organism. Signs of tetanus occur from about 4 days to 3 weeks or longer after infection is established in a wound. The animal may have a stiff gait, lockjaw can develop, and the third eyelid may protrude across the eye. The animal usually will go down with all four legs held out straight and stiff and the head drawn back. Convulsions may occur.
Abortion Diseases
Ewes or does that lose their lambs early in pregnancy may not return to heat because they are seasonal breeders, and so may not be bred back that season. This is a significant loss for the producer both because of decreased lamb crop and because those dams will not lactate that season. Pregnancy also may be lost late in gestation or weak or deformed lambs may be born. For many diseases causing abortion in small ruminants, there is no vaccine in the United States (for example, Q fever, toxoplasmosis, Border disease). In the United States, the most common causes of abortion in ewes are Chlamydia / Chlamydophila (enzootic abortion) and Campylobacter. Enzootic abortion is transmitted from aborting dams to other females in the flock. Ewe lambs are the most susceptible on farms where the organism is present. Abortion usually is seen late in pregnancy or lambs may be born that die shortly after birth. Campylobacteriosis (formerly called Vibriosis) causes abortion late in pregnancy or birth of stillborn or weak lambs. Infecting organisms are Campylobacter jejuni and Campylobacter fetus. Ewes are infected orally and the incubation time from ingestion to abortion is just 2 weeks.
Contagious Ecthyma (soremouth, orf, scabby mouth, or pustular dermatitis)
Soremouth is the most common skin disease affecting sheep and goats. It is a highly contagious viral infection that also can produce painful infections in humans. The virus causes scab formation on the skin, usually around the mouth, nostrils, eyes, mammary glands, and vulva. It first appears as tiny red nodules, usually at the junctions of the lips.
Caseous Lymphadenitis (CLA, boils, abscesses, or cheesy gland)
Caseous lymphadenitis is an infectious, contagious disease that primarily infects the lymphatic system, though other organs can be affected. It is caused by Corynebacterium pseudotuberculosis. Infection results in abscess formation in the lymph nodes that when cut or ruptured, discharge pus containing the bacteria in the surroundings. When the infection spreads internally, affected animals slowly lose weight and eventually become emaciated.
Vaccines for Disease in Sheep and Goats
Specific vaccines that are available are:
- CD-T (Clostridium perfringens types C and D and tetanus toxoid)
- Abortion vaccines
- Contagious ecthyma (soremouth, orf)
- Caseous lymphadenitis (CLA)
- Rabies
CD-T (Clostridium perfringens types C and D and tetanus toxoid)
Protection against type C is needed by nursing lambs and kids on all farms, regardless of management practices. Maternal antibodies will be present in colostrum if the ewe/doe was vaccinated in the 2-4 weeks prior to parturition. This passive immunity lasts in the lamb/kid for their first 50-60 days of life. Protection against type D is needed in lambs/kids fed grain or lush forage (any high-carbohydrate diet). It is associated with production of a toxin that is acutely fatal in fast-growing lambs. Tetanus toxoid is included in this product. Ewes and does should be vaccinated 8 weeks prior to the first time they give birth, and boostered at 2-4 weeks prior to parturition. Previously vaccinated ewes and does only require the vaccination 2-4 weeks prior to parturition. Offspring from vaccinated dams are vaccinated at weaning and again 3-4 weeks later. Offspring from unvaccinated dams are vaccinated as newborns (may have systemic reaction, can be treated with epinephrine) and are boostered at 3-4 weeks of age and again at weaning. All animals, including males, should be boostered at least annually; some goats may only maintain protective antibody titers for six months after vaccination.
Abortion Vaccines
In general, do not vaccinate if there are no problems on the farm or if animals are determined to be at low risk. Risk is associated with having an open flock, with animal traffic on and off the farm, with free-roaming wildlife, feeding on the ground, or with a history of abortions in the flock. Diagnosis for abortion problems is difficult and for many diseases causing abortion in small ruminants, there is no vaccine in the United States (for example, Q fever, toxoplasmosis, Border disease).
- Chlamydia / Chlamydophila (enzootic abortion) vaccine may not be effective against all strains causing abortions and regional variations in protection may exist. The label states that the vaccine should be administered 60 days prior to breeding with a booster in 30 days.
- Campylobacter (formerly called Vibrio) vaccination induces immunity for only 4-5 months. The label states that the first dose of vaccine should be given 2 weeks prior to breeding and that it should be boostered 2-3 months later. In the face of an “abortion storm,” where many abortions are occurring in the flock due to this organism, revaccination of all animals may reduce losses.
Contagious Ecthyma (soremouth, orf)
This is a live virus vaccine that will introduce a mild infection. This vaccine should only be used if this disease is present in the flock. The vaccine is given percutaneously; an area of skin in a wool-less region (inside the thigh or ear, or under the tail) is scarified in the form of an X deeply enough to cause inflammation but not so deeply as to cause bleeding. The vaccine is brushed on. The vaccinated area will scab over and does contain live virus that will be infective even after falling off the sheep. Vaccination under the tail is preferred to that inside the thigh, as vaccination on the medial thigh region may cause irritation and scabbing over the mammary glands and teats. This is a zoonotic disease so people should wear gloves and exercise caution when handling the vaccine or sheep, or picking up scabs. On farms that have a problem with this disorder, each new lamb and kid crop should be vaccinated. Lambs moving into feedlots should be vaccinated at least 14 days before shipment. Do not use this vaccine on farms that do not already have this disorder.
Caseous Lymphadenitis (CLA)
This may be a stand-alone vaccine or combined with CD-T vaccine. Vaccination decreases severity of disease but does not prevent disease. The vaccine should not be used in flocks or herds unless they have been exposed or contain affected individuals.
Rabies
This is a killed vaccine. Several products are available for sheep. No products are specifically labeled for goats but there is a recommendation in some situations that publicly displayed goats will be vaccinated. For example, goats take to a show that will be housed in a pen probably will not be vaccinated for rabies, while goats at a petting zoo that are purposefully brought to that site to interact with people will be vaccinated for rabies. Animals are vaccinated when greater than 3 months of age and then annually.

*
 SWINE
SWINE
Vaccination is a common preventive medicine practice on commercial swine farms. Vaccination protocols for swine typically focus upon preventing diseases of the reproductive, respiratory, and gastrointestinal tracts, and preventing multi-systemic disease. Vaccines may be applied to the reproductive herd (sows and boars), replacement females (gilts), or growing pigs, depending on when the disease challenge is expected.
Forms of Vaccination Production
- Commercial (manufactured and sold by pharmaceutical companies)
- Autogenous (typically manufactured by a licensed laboratory from organisms isolated from the farm of concern)
- On Farm – Typically includes other, less controlled, methods that stimulate immunity such as oral feeding of infectious organisms (‘biofeedback’) or inoculation with serum from viremic animals (serum inoculation)
Routes of administration for swine vaccines typically include IM or oral (per os = PO). Multivalent vaccines (those containing multiple organisms) are commonly created to decrease the stress, labor, and food safety issues associated with multiple injections. Swine farm clientele expect that veterinarians will consider the costs and benefits associated with the vaccination protocols they recommend.
Pork Quality Assurance Vaccination Recommendations
Pork Quality Assurance programs strongly encourage the use of vaccines to prevent the necessity of antibiotic therapy and the potential for post therapeutic residues. They recommend the following when administering vaccines to pigs:
- Use a spot on the neck just behind and below the ear, but in front of the shoulder. Do not use a needle to inject in the ham or loin. There may be some bleeding and bruising of the muscle followed by scarring. This scar can stay in the muscle for the life of the pigs and be a blemish in the cut of meat. This standard applies to sows as well as market hogs. While sows may not be going to market soon, they are at greater risk for blemishes because of the repeated injections they typically receive over their productive life in the form of vaccinations and farrowing medications.
- Use the proper size and length of needle to ensure the medication is deposited in the muscle, not in other tissues.
| GAUGE OF NEEDLE | LENGTH OF NEEDLE (INCHES) | |
| Baby Pigs | 18 or 20 | ½ or ⅝ |
| Nursery Pigs | 16 or 18 | ⅝ or ¾ |
| Finisher Pigs | 16 | 1 |
| Breeding Stock | 14, 15 or 16 | 1 or 1½ |
Diseases in Swine
Diseases to vaccinate against in swine include:
- Leptospirosis
- Erysipelas
- Porcine parvovirus (PPV)
- Porcine Reproductive and Respiratory Syndrome (PRRS)
- Mycoplasma hyopneumoniae
- Influenza
- Escherichia coli
- Rotavirus
- Porcine Proliferative Enteritis (PPE)
- Porcine Circovirus Type 2 (PCV2)
- Meningitis
Leptospirosis
As in cattle, leptospirosis is a zoonotic disease, caused by bacteria of the genus Leptospira. There are many different serotypes of disease and prevalence varies with geographic location. Worldwide, pigs are the maintenance hosts for pomona, tarassovi, bratislava, and muenchen. Pomona causes important reproductive problems in breeding sows, spreading slowly through the herd. The skunk is a reservoir host. Once the organism is introduced into a herd, the pigs become permanent carriers with infection of the kidneys and intermittent excretion of the organism into the urine. Piglets rarely are infected. The most common manifestation is chronic low-grade disease in sows, with abortions, stillbirths, and birth of weak piglets.
Erysipelas
Swine erysipelas is caused by a bacterium, Erysipelothrix rhusiopathiae, that is found on most if not all pig farms. Up to half of the animals on a farm may carry it in their tonsils. It is excreted in saliva, feces, and urine, and so is common in the environment. It is also found in many other species, including birds, and can survive for long periods in the environment. The bacterium alone may cause disease but clinical disease is more common if there is concurrent infection with viral diseases such as porcine reproductive and respiratory syndrome (PRRS) virus and influenza. Disease is relatively uncommon in pigs under 8-12 weeks of age due to protection provided by maternal antibodies from colostrum. The most susceptible animals are growing pigs, non-vaccinated gilts, and young sows. Infected sows may show acute death in apparently healthy animals, fever, abortion, stillbirths, birth of mummified piglets, raised areas in the skin (“diamonds”) that turn red and then black, or joint stiffness. Infertility may be a presenting concern on a farm. Infected boars with high fevers have transient decrease in semen quality, reflected as sows not getting pregnant or having smaller litters. Infected growing pigs may show acute death, fever, and the characteristic skin lesion described above.
Porcine Parvovirus (PPV)
PPV is the most common and important cause of infectious infertility in pigs. Porcine parvovirus multiplies normally in the intestine of the pig without causing clinical signs. It is worldwide in its distribution. PPV can persist in the environment for many months and is resistant to most disinfectants. Infected sows show only reproductive signs of disease. Parvovirus is associated with lack of conception, birth of mummified or stillborn pigs, and birth of live pigs with low birth weight. Sporadic disease is seen in individual females that are infected for the first time; for this reason, disease usually is seen in gilts. Once a pig is exposed, it has lifelong immunity. Reproductive problems in a herd appear about every 3-4 years if vaccination is not practiced.
Porcine Reproductive and Respiratory Syndrome (PRRS)
This is a fairly new disease; it has only been recognized in the United States since the mid-1980s and was only identified as being caused by an arterivirus in 1991. The virus has an affinity for macrophages, especially those in the lung. Up to 40% of macrophages are destroyed in a given animal, making it susceptible to other diseases. When introduced into a new herd, 90% of breeding sows will be seropositive within 4-5 months. Grower pigs shed virus for months; adult pigs shed virus for periods of time as short as 2 weeks. Nasal secretions, saliva, feces, and urine may contain virus. The virus can be airborne for up to 2 miles and can be carried between pigs and on fomites (inanimate objects) including boots, equipment, and trucks. The virus also may be carried by flies and mosquitoes. The clinical picture varies tremendously from one herd to another. As a guide, for every three herds that are exposed to PRRS for the first time, one will show no recognizable disease, the second will show mild disease, and the third will show moderate to severe disease. The reasons for this are not clearly understood. Clinical signs in sows include inappetance; fever; abortions; early farrowing with birth of mummified, stillborn, or weak pigs; prolonged return to heat after weaning of piglets; coughing; lack of milk and mastitis; lethargy; and cyanosis (blue discoloration) of the ears. This is the acute phase of the disease. Long-term, reproductive efficiency in herds in which the infection has become enzootic are associated with 10-15% reduction in farrowing rate, increased stillbirths, increased number of abortions, and inappetance in sows at farrowing. Infected piglets may show diarrhea and increase in other respiratory infections. Infected weaning and growing pigs may show other infections secondary to PRRS infection with a variety of clinical signs and mortality rate as high as 12-15%.
Mycoplasma hyopneumoniae
This is one of the most important contributors to respiratory disease in pigs. The organism damages the cilia and epithelia of the airways of the lower respiratory tract, permitting infection with other organisms, for example PRRS virus. Mycoplasma is transmitted via direct contact with infected pigs. Pigs older than 6 weeks are mainly those affected. Clinical signs include a non-productive cough, rough hair coat, and reduced growth rate and feed efficiency. With secondary bacterial infection, signs are more severe and include labored breathing, a harsher cough, fever, and prostration.
Influenza
Influenza viruses are the cause of outbreaks of acute respiratory disease. Influenza A viruses infect a wide range of avian and mammalian species, with the latter group including humans, pigs, horses, and aquatic mammals. Type A viruses are known for their ability to change their antigenic structure and create new strains. The type A viruses are further divided into serotypes, based on the antigenic nature of their surface glycoproteins hemagglutinin (H) and neuraminidase (N). Swine influenza in large herds may become endemic with intermittent bouts of disease and infertility. Different influenza strains may sequentially infect the herd. Immunity to influenza viruses is often short-lived (6 months). Outbreaks are seen throughout the year. Piglets generally are protected by maternal antibodies they receive in colostrum. Sows may show pregnancy loss secondary to fever and coughing. Weaner and grower pigs with acute illness classically are fine one day and then lying prostrate and breathing heavily the next.
Escherichia coli
Diarrhea is the most common and important disease of piglets. In a well-managed herd, there should be fewer than 3% of litters at any time requiring treatment for diarrhea and piglet mortality from diarrhea should be less than 0.5%. At birth, the gastrointestinal tract is microbiologically sterile. Organisms begin to colonize the tract quickly after birth, among them potentially pathogenic strains of E. coli. Ingestion of colostrum and later, of milk containing IgA, is vital for creation of immunity within the intestinal tract. If too many bacteria are present or if there are stressors present such as chilling or concurrent infection, piglets will succumb to disease. Periods of greatest risk are before 5 days of age and between 7 and 14 days of age. Weaning is another risk, as loss of sow’s milk and the IgA it contains allows the bacteria to attach to the villi of the small intestines. Signs may include acute death, dehydration, and sticky feces around the rectum and tail with an accompanying characteristic sour smell.
Rotavirus
This virus is widespread in pig populations, with virtually 100% seroconversion in adult stock. It is resistant to environmental changes and many disinfectants and so persists for long periods of time in the environment. Piglets are initially protected from maternal antibodies in colostrum but become susceptible to infection by about 3-6 weeks of age. Exposure does not necessarily result in disease; it is estimated that only 10-15% of diarrheas in pigs are due to primary rotavirus infection. The virus destroys the intestinal villi, preventing fluid uptake and causing watery diarrhea.
Porcine Proliferative Enteritis (PPE)
This is a disease characterized histologically by inflammation, ulceration, and hemorrhage in the intestinal tract. The causative organism, Lawsonia intracellularis, is a unique obligate intracellular organism related to anaerobic bacteria. Clinical disease is characterized as an acute form common in young adults and a chronic or necrotic form in grower pigs. Carrier animals shed the organism in their feces and susceptible pigs are exposed through the fecal-oral route. Carrier sows may infect nursing pigs as early as 6 days of age. Pigs present with pallor, weakness, and rapid death. Subacute to chronic cases occur more frequently in grower pigs, which show sporadic diarrhea, wasting, and variation in growth rate.
Porcine Circovirus Type 2 (PCV2)
This is a widespread virus and essentially all pig herds are infected. However, very few have PCV2-associated disease which can include a variety of systemic disorders including clinical signs of wasting, pneumonia, enteritis, and reproductive failure. The hallmark of PCV2 infection is depletion or inflammation of lymphoid tissue. In many cases, PCV2 infection requires a trigger such as concurrent infection (PRRS virus, Mycoplasma hyopneumoniae) or other stressors.
Meningitis
Hemophilus parasuis and Streptococcus suis are two organisms associated with inflammation of the meninges in pigs. Signs of meningitis in piglets and weaned pigs include rapid onset of recumbency, shivering, nystagmus (shaking of the eyeballs), paddling, and convulsions. In older pigs (growers and adults), muscle trembling, nystagmus, and incoordination are more common clinical signs.

Vaccines for Disease in Swine
Vaccination for Reproductive Disease
Common vaccinations include:
- Leptospirosis-Erysipelas-Parvovirus combination vaccine
- Each of these components may be administered as individual vaccinations as well
- Porcine Reproductive and Respiratory Syndrome virus (PRRS) vaccine
- Modified live virus vaccine typically thought to be most effective
Common uses are:
- Typically administered twice prior to the first breeding of gilts and once before subsequent gestation periods in reproductive herd (sows and boars)
Vaccination for Respiratory Disease
Common vaccinations include:
- Mycoplasma hyopneumoniae vaccine
- Single dose and two dose versions exist
- Swine Influenza virus vaccine
- Normally include H1N1 and H3N2 components
- Porcine Reproductive and Respiratory Syndrome virus (PRRS) vaccine
Common uses are:
- Routine vaccinations in reproductive herds to maintain respiratory disease stability
- Acclimation of disease-negative replacement females as they enter disease-positive herds
- Growing pigs at weaning or during the nursery phase to prevent significant respiratory disease during the finisher phase
Vaccination for Gastrointestinal Disease
Common vaccinations include:
- Escherichia coli vaccine
- Multiple strains typically included in the vaccine
- Rotavirus vaccine
- Modified live virus, only one type included in commercial vaccine
- Lawsonia intracellularis vaccine
- A modified live vaccine that requires storage at -70°C and it administered orally following thawing and dilution
Common uses are:
- Escherichia coli and Rotavirus vaccines commonly used prior to farrowing to prevent neonatal scours in suckling piglets
- Lawsonia intracellularis vaccines administered in late nursery or early finisher (9-12 weeks of age) to prevent ileitis in finisher pigs OR for the acclimation of replacement females into breeding herds
Vaccination for Multi-Systemic Disease
Common vaccinations include:
- Porcine Circovirus Type 2 (PCV2) vaccine
- Currently the most common vaccine used in swine
- Considered to be highly effective in preventing a devastating disease syndrome
- Evidence that it provides effective protection in both single and two dose programs.
- Hemophilus parasuis vaccine
- Typically includes multiple serotypes that provide cross protection for other serotypes
- Erysipelas rhusiopathiae vaccine
- May be delivered IM or PO
- Streptococcus suis vaccine
- Efficacy is questionable
- Commonly produced as an autogenous vaccine
Common uses are:
- These vaccines are often manufactured in combination type products with respiratory specific vaccines to prevent disease in replacement females or growing pigs.


