Proteins: amino acids
Learning Objectives
Know what an amino acid is. Know how many amino acids there are.
Know that different proteins have different amino acids (e.g., albumin in eggs is one of the few “complete” proteins).
Hey everyone today we’re going to be learning about the building blocks, structures and roles of Proteins. Focusing heavily on amino acids.
Proteins are among the most abundant organic molecules in living systems and are way more diverse in structure and function than other classes of macromolecules. A single cell can contain thousands of proteins, each with a unique function. Although their structures, like their functions, vary greatly, all proteins are made up of one or more chains of amino acids.
(pull up amino acid figure that’s below)
Amino acids are the monomers that make up proteins. A protein is made up of one or more linear chains of amino acids, each of which is called a polypeptide. There are 20 types of amino acids commonly found in proteins.
We’ll come back to amino acids and polypeptides, I just wanted to get you thinking of them for a second and to know that there’s 20 commonly found.
Right now we’re going to quickly go over enzymes and hormones,
Enzymes act as catalysts in biochemical reactions, meaning that they speed the reactions up. Each enzyme recognizes one or more substrates, the molecules that serve as starting material for the reaction it catalyzes. Different enzymes participate in different types of reactions and may break down, link up, or rearrange their substrates.
One example of an enzyme found in your body is salivary amylase, which breaks amylose (a kind of starch) down into smaller sugars. Amylose doesn’t taste very sweet, which is why starchy foods often taste sweeter if you chew them for longer: you’re giving salivary amylase time to get to work.
One more example of an enzyme is lactase. People who are dairy intolerant, or lactose intolerant, can’t digest lactose because their body doesn’t produce the enzyme, lactase. –side note, If you do have this issue, there’s over the counter supplements that one can take before eating dairy that has lactase added to it, making it possible to consume dairy without the side effects.
Now we’re going to move onto hormones. Hormones are long-distance chemical signals released by endocrine cells (like the cells of your pituitary gland). They control specific physiological processes, such as growth, development, metabolism, and reproduction. While some hormones are steroid-based, others are proteins, commonly called peptide hormones.
For example, insulin is an important peptide hormone that helps regulate blood glucose levels. When blood glucose rises (for instance, after you eat a meal), specialized cells in the pancreas release insulin. The insulin binds to cells in the liver and other parts of the body, causing them to take up the glucose. This process helps return blood sugar to its normal, resting level.
A protein’s shape is critical to its function, many different types of chemical bonds may be important in maintaining this shape. Changes in temperature and pH, as well as the presence of certain chemicals, may disrupt a protein’s shape and cause it to lose functionality, a process known as denaturation.
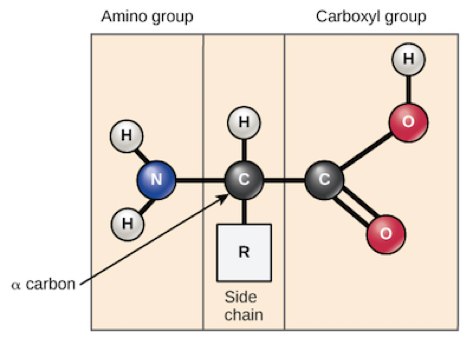
Coming back to amino acids, as you look at this figure, know this is the basic amino acid structure, consisting of a central carbon atom, also known as the alpha (α) carbon, bonded to an amino group(NH2), a carboxyl group (COOH), as well as a hydrogen atom.
Now this is not actually the state in which an amino acid would typically be found. At physiological pH, which is (7.2-7.4), the amino group is typically protonated and bears a positive charge, while the carboxyl group is typically deprotonated and bears a negative charge. Each amino acid has another atom, or group of atoms, bonded to the central atom, known as the R group. This determines the identity of the amino acid.
(here’s a figure to quickly glance at to jog the memory of pronation vs deprotonation)
https://pediaa.com/difference-between-protonation-and-deprotonation/
One example of an amino acid is that if the R group is a hydrogen atom, then the amino acid is glycine, while if it’s a methyl (CH3) group, the amino acid is alanine. The 20 common amino acids are shown in this chart.
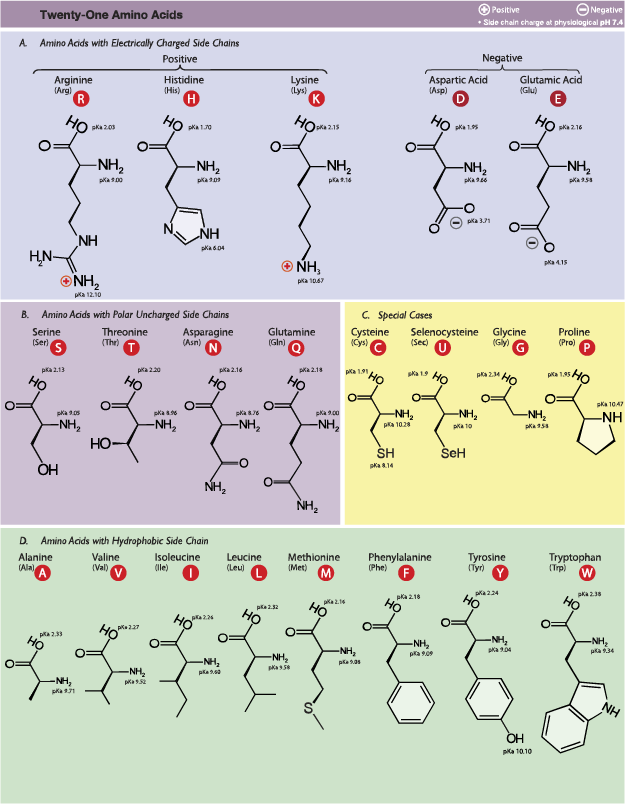
The properties of the side chain determine an amino acid’s chemical behavior (that is, whether it is considered acidic, basic, polar, or nonpolar). For example, amino acids such as valine and leucine are nonpolar and hydrophobic, while amino acids like serine and glutamine have hydrophilic side chains and are polar. Some amino acids, such as lysine and arginine, have side chains that are positively charged at physiological pH and are considered basic amino acids. Aspartate and glutamate, on the other hand, are negatively charged at physiological pH and are considered acidic.
Now, we’re moving onto Peptide Bonds. Each protein in your cells consists of one or more polypeptide chains. Each of these polypeptide chains is made up of amino acids, linked together in a specific order. A polypeptide is kind of like a long word that is “spelled out” per se, in amino acid letters. The chemical properties and order of the amino acids are key in determining the structure and function of the polypeptide, and the protein it’s part of. But how are amino acids actually linked together in chains you may ask? -Khan/words added
The amino acids of a polypeptide are attached to their neighbors by covalent bonds known as peptide bonds. Each bond forms in a dehydration synthesis, or (condensation), reaction. During protein synthesis, which will be talked about in another lecture, the carboxyl group of the amino acid at the end of the growing polypeptide chain reacts with the amino group of an incoming amino acid, releasing a molecule of water. The resulting bond between amino acids is a peptide bond
You’ll see here how the water molecule, highlighted in red, bonds the two amino groups resulting in the connection shown.
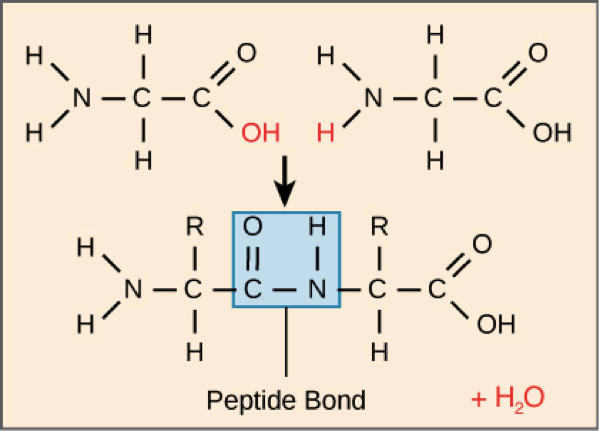
Amino acid figure from Biology. Provided by: OpenStax CNX. Located at: http://cnx.org/contents/185cbf87-c72e-48f5-b51e-f14f21b5eabd@10.8. License: CC BY: Attribution. License Terms: Download for free at http://cnx.org/contents/185cbf87-c72e-48f5-b51e-f14f21b5eabd@10.8

